Breakthrough reinforces biotech firm’s leadership in regenerative medicine



Longeveron Inc. has secured a major intellectual property milestone in China, reinforcing its global strategy in regenerative medicine. The clinical-stage biotechnology company announced that China’s patent authority has granted it protection covering advanced methods for evaluating stem cell potency—an essential component in the development of safe and effective cell therapies.
This latest patent strengthens the company’s expanding international portfolio and positions it competitively in one of the world’s fastest-growing biotechnology markets.
Patent Covers Critical Stem Cell Testing Technology
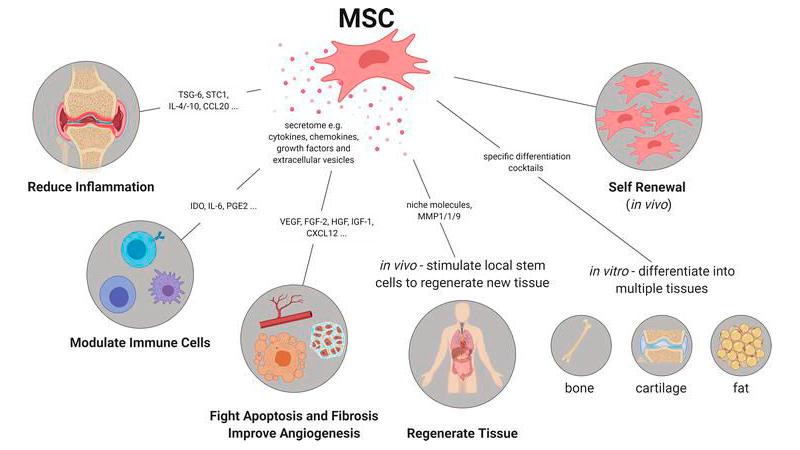
The newly granted patent protects potency assay methods used to assess human mesenchymal stem cells (MSCs)—a cornerstone of modern regenerative therapies.
These assays determine whether stem cells possess the biological activity required to deliver therapeutic benefits. Without such validation, regulatory approval for cell-based therapies becomes significantly more difficult.
The patent applies to MSCs derived from multiple biological sources, including:
- Bone marrow
- Adipose (fat) tissue
- Peripheral blood
- Umbilical cord and placenta
- Amniotic membrane and fluid
- Induced pluripotent stem cells (iPSCs)
This broad scope enhances the patent’s commercial and clinical value, as it covers a wide range of cell sources commonly used in regenerative medicine.
Protection Valid Through 2041
The Chinese patent—titled “Potency Assay”—provides Longeveron with exclusive rights in the country through 2041, subject to standard maintenance requirements.
This long-term protection offers a strategic advantage in China’s rapidly evolving biotech ecosystem, where demand for advanced therapies is accelerating due to aging populations and increasing chronic disease burden.
By securing rights in China, Longeveron not only protects its innovations but also creates opportunities for:
- Licensing agreements
- Strategic partnerships
- Future commercialization pathways
Strategic Expansion of Global Patent Portfolio
The China patent adds to Longeveron’s growing global intellectual property portfolio, which now includes more than 50 issued patents worldwide.
Company leadership emphasized the importance of IP strength in scaling regenerative medicine technologies. The expanding patent base represents a critical strategic asset that supports the company’s long-term mission of delivering innovative therapies to patients.
This move also aligns with Longeveron’s broader trend of securing patents across key international markets, reflecting a coordinated global IP expansion strategy.
Why Potency Assays Matter in Regenerative Medicine





Potency assays are not just a technical requirement—they are a regulatory necessity.
In cell therapy development, these assays:
- Verify biological activity of living cells
- Ensure consistency between manufacturing batches
- Predict therapeutic effectiveness
- Support regulatory approvals
Without reliable potency testing, even promising therapies can fail to reach commercialization.
Longeveron’s patented methods aim to standardize and improve this critical step, potentially accelerating the approval timeline for its therapies.
Flagship Therapy Laromestrocel Gains Momentum
The patent directly supports Longeveron’s lead investigational product, laromestrocel (Lomecel-B™)—a mesenchymal stem cell therapy derived from healthy adult donors.
This therapy is currently under investigation for multiple high-impact conditions, including:
- Hypoplastic Left Heart Syndrome (HLHS)
- Alzheimer’s disease
- Aging-related frailty
- Pediatric dilated cardiomyopathy
Clinical progress has been notable. The therapy has already received several key regulatory designations in the United States, highlighting both the urgency of the targeted conditions and the therapy’s potential.
Phase 2b Trial Could Be Pivotal
Longeveron is currently conducting a Phase 2b clinical trial evaluating laromestrocel for HLHS—a rare and life-threatening pediatric heart condition.
The company expects trial results in Q3 2026, which could mark a turning point.
If successful, the data may support a regulatory filing with U.S. authorities, potentially bringing the therapy closer to commercialization.
This makes the newly granted patent even more critical, as it strengthens the regulatory and commercial foundation ahead of potential approval.
China Patent Signals Market Intent
China represents a massive opportunity for regenerative medicine companies. With:
- A rapidly aging population
- Rising healthcare spending
- Strong government support for biotech innovation
the country has become a strategic priority for global life sciences firms.
By securing patent protection in China, Longeveron signals clear intent to participate in this high-growth market. The move also helps safeguard the company against potential IP risks in a competitive landscape where innovation cycles are accelerating.
Competitive Positioning in a High-Stakes Sector
The global regenerative medicine market is intensifying, with biotech firms racing to develop scalable, effective therapies for chronic and rare diseases.
Longeveron’s approach—combining:
- A strong clinical pipeline
- A robust patent portfolio
- Strategic geographic expansion
positions it as a serious contender in the space.
Its focus on mesenchymal stem cells, known for their regenerative and anti-inflammatory properties, further enhances its differentiation.
Outlook: Strong IP Backbone Supports Future Growth
Longeveron’s China patent marks more than a legal win—it reflects a strategic consolidation of scientific innovation and commercial foresight.
With key clinical milestones approaching and a growing IP portfolio, the company is steadily building the infrastructure needed to transition from a clinical-stage biotech to a commercial-stage player.
If upcoming trial results align with expectations, Longeveron could emerge as a significant force in regenerative medicine—backed by a patent portfolio designed to protect and scale its innovations globally.