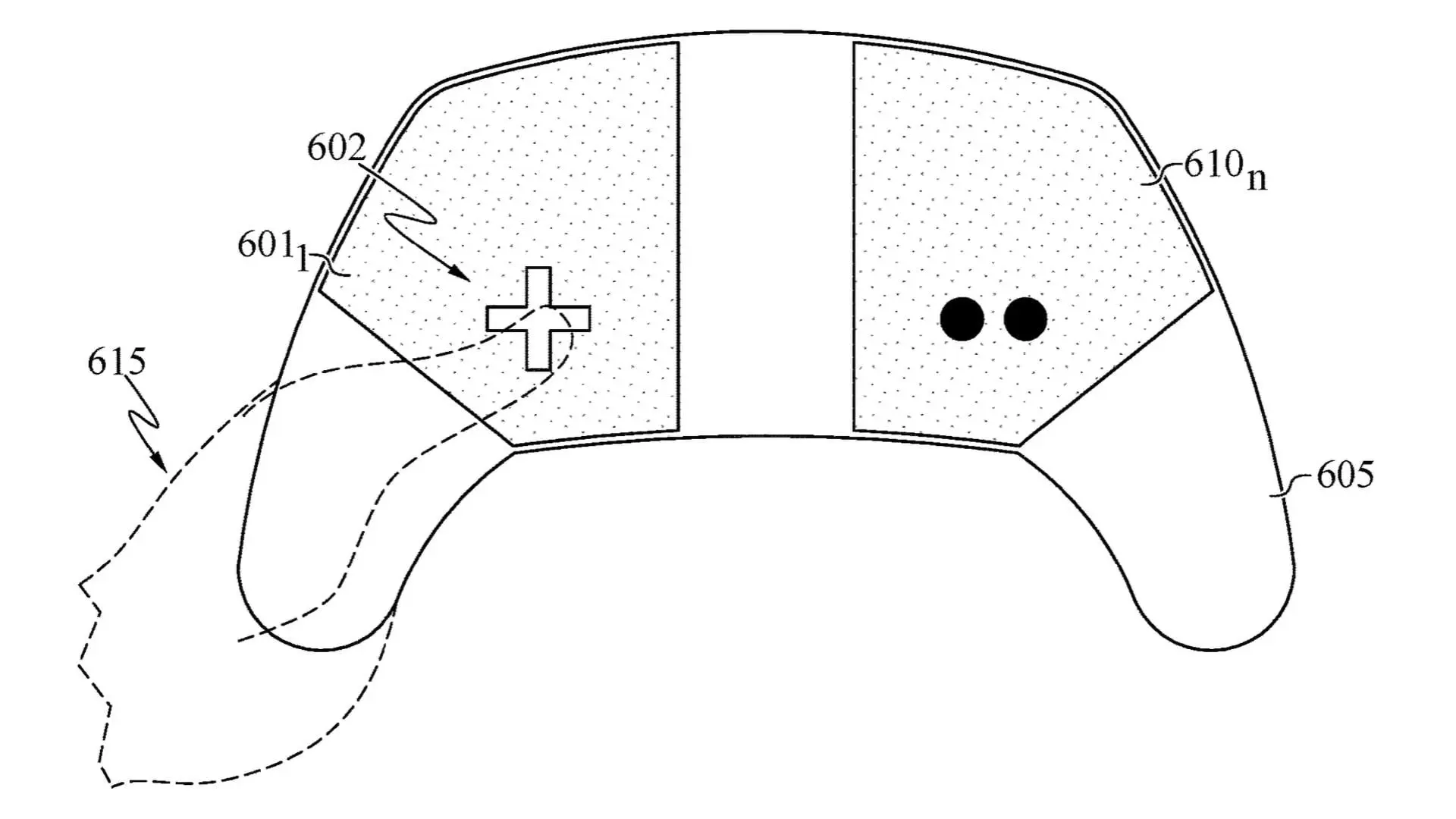
The global technology industry is witnessing another major legal confrontation as Swedish telecom giant Ericsson has filed a lawsuit against Taiwanese electronics manufacturer Acer over alleged disputes involving 4G and 5G wireless patent licensing. The case highlights rising tensions over standard-essential patents (SEPs) and underscores how licensing conflicts are reshaping the competitive landscape of next-generation connectivity.
The lawsuit, filed in a United States federal court, centers on claims that Acer failed to comply with industry-standard licensing obligations and pursued aggressive litigation strategies against telecom operators using Ericsson’s network equipment. The legal clash adds to a growing wave of high-stakes patent disputes that increasingly define relationships between infrastructure providers, device manufacturers, and telecom carriers worldwide.
A Strategic Legal Move by Ericsson
Ericsson’s legal action seeks clarity and protection against what the company describes as escalating threats from Acer’s patent enforcement tactics. According to court filings, Ericsson is requesting a judicial declaration confirming that it does not infringe Acer’s patents and that Acer violated obligations to negotiate licensing agreements under fair terms.
The Swedish telecom company argues that Acer pursued litigation against Ericsson’s customers rather than engaging constructively in licensing negotiations. Such actions, Ericsson claims, disrupt industry norms and undermine the cooperative framework designed to ensure interoperability across global telecom standards.
By filing the lawsuit, Ericsson aims to shift the legal battleground away from indirect disputes involving network operators and toward a direct resolution between patent holders.
The Role of Standard-Essential Patents
At the heart of the dispute lies the complex world of standard-essential patents. These patents cover technologies required to implement globally recognized wireless standards such as 4G LTE and 5G.
Companies that hold SEPs commit to licensing them under FRAND principles — fair, reasonable, and non-discriminatory terms. The FRAND framework exists to balance innovation incentives with industry accessibility. Without such rules, companies could block competitors from using critical technologies needed to maintain interoperable networks.
Ericsson claims Acer’s actions conflict with these obligations. The lawsuit alleges that Acer adopted strategies that pressure telecom operators and ecosystem partners instead of resolving licensing issues through direct negotiation.
Dispute Escalation: From Negotiations to Litigation
Industry observers note that patent licensing disagreements often begin with negotiations over royalty rates or usage rights. In this case, however, talks reportedly broke down, triggering a chain reaction of legal actions.
Acer allegedly filed infringement claims against major telecom operators, including U.S. carriers that deploy Ericsson equipment. Ericsson argues that targeting its customers indirectly places pressure on its business relationships and could disrupt ongoing network operations.
The lawsuit seeks to halt what Ericsson views as a strategy designed to extract licensing concessions by threatening key industry partners.
This escalation reflects a broader trend. Instead of pursuing bilateral licensing discussions, companies increasingly resort to multi-jurisdictional litigation to strengthen bargaining positions.
Comparative Landscape: Device Makers vs Infrastructure Providers
The Ericsson-Acer dispute illustrates the evolving dynamics between device manufacturers and telecom infrastructure vendors.
Traditionally, network equipment providers like Ericsson focused on supplying infrastructure to operators, while consumer device companies negotiated patent licenses separately. However, the boundaries between these roles have blurred as companies diversify portfolios and accumulate extensive patent holdings.
In recent years, technology firms have leveraged intellectual property as both defensive shields and offensive tools. Companies with significant patent portfolios can exert influence across multiple segments of the ecosystem, from handsets and laptops to network hardware and cloud platforms.
This shift has led to more frequent clashes between entities that previously operated in largely separate spheres.
Why FRAND Compliance Matters
FRAND licensing serves as a cornerstone of modern telecommunications. Without it, industry collaboration on shared standards would be nearly impossible.
Standards organizations require patent holders to commit to fair licensing practices to prevent monopolistic behavior. Yet disagreements often arise over what constitutes “fair” or “reasonable” compensation.
Legal disputes frequently hinge on:
- Royalty calculation methods
- Geographic licensing scope
- Portfolio valuation
- Negotiation conduct and timelines
Ericsson’s lawsuit emphasizes negotiation behavior rather than solely technical infringement claims. This strategic framing could influence how courts evaluate the balance between enforcement rights and industry cooperation.
Potential Industry Impact
The outcome of the lawsuit could shape future licensing negotiations across the telecom sector. If courts support Ericsson’s position, companies may face stricter expectations to negotiate directly and avoid targeting downstream customers as leverage.
Conversely, a ruling favorable to Acer could reinforce aggressive enforcement strategies by patent holders, encouraging similar litigation tactics across the industry.
The dispute also arrives at a critical moment for 5G deployment. Telecom operators continue investing heavily in infrastructure upgrades, and prolonged legal uncertainty could complicate procurement decisions and supply chain stability.
A Broader Pattern of Telecom Patent Battles
The Ericsson-Acer case is not isolated. Telecom companies frequently engage in complex patent disputes as the industry transitions toward advanced wireless technologies.
Over the past decade, similar legal confrontations have involved major players across Europe, Asia, and North America. Companies increasingly seek judicial clarification on FRAND obligations, jurisdictional authority, and cross-licensing frameworks.
Experts say these conflicts reflect the enormous financial stakes associated with 5G innovation. With billions invested in research and development, patent holders aggressively protect their intellectual property while implementers push for predictable licensing costs.
Market and Legal Implications
Beyond legal arguments, the lawsuit carries strategic implications for both companies.
For Ericsson, defending its relationships with telecom operators remains a priority. By seeking court intervention, the company aims to prevent disruptions that could affect customer confidence and network deployment timelines.
For Acer, asserting patent rights signals its intent to expand influence within the wireless technology ecosystem. As device manufacturers diversify into connected hardware and enterprise solutions, control over key patents becomes a powerful competitive advantage.
Investors and industry stakeholders will closely monitor the case for signals about licensing trends and legal risk exposure across the sector.
What Comes Next
The court proceedings will likely involve detailed technical analysis, economic modeling, and examination of negotiation history. Patent litigation often spans months or years, especially when multiple jurisdictions and complex licensing frameworks are involved.
Regardless of the outcome, the dispute reinforces a central reality of the modern telecom industry: innovation and litigation increasingly move hand in hand.
As companies race to define the future of connectivity through 5G and beyond, intellectual property battles will continue to shape alliances, market strategies, and the pace of technological adoption.
For now, Ericsson and Acer find themselves at the center of a high-profile legal contest that could redefine how patent licensing conflicts unfold in the era of global wireless standards.